What Is the Amount of Heat Required to Raise the Temperature of 200.0 G of Aluminum by 10ã‚â°c
Chapter 5. Thermochemistry
v.2 Calorimetry
Learning Objectives
By the end of this section, you will be able to:
- Explicate the technique of calorimetry
- Calculate and interpret rut and related properties using typical calorimetry information
One technique we can use to measure the corporeality of heat involved in a chemical or physical process is known equally calorimetry. Calorimetry is used to measure out amounts of rut transferred to or from a substance. To do so, the rut is exchanged with a calibrated object (calorimeter). The change in temperature of the measuring part of the calorimeter is converted into the corporeality of heat (since the previous calibration was used to establish its oestrus capacity). The measurement of heat transfer using this approach requires the definition of a organisation (the substance or substances undergoing the chemical or physical change) and its environment (the other components of the measurement apparatus that serve to either provide heat to the system or absorb oestrus from the system). Knowledge of the heat capacity of the surroundings, and careful measurements of the masses of the system and surroundings and their temperatures before and later the process allows one to calculate the estrus transferred every bit described in this section.
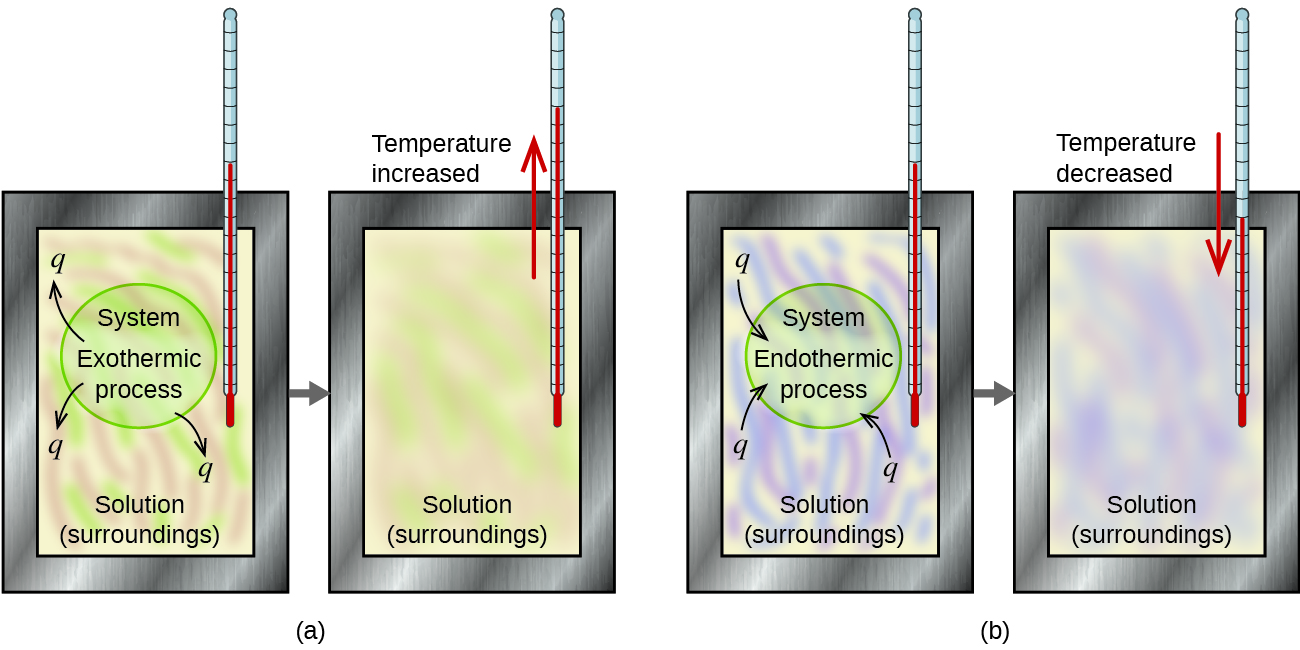
A calorimeter is a device used to measure the amount of oestrus involved in a chemic or physical process. For example, when an exothermic reaction occurs in solution in a calorimeter, the heat produced by the reaction is captivated by the solution, which increases its temperature. When an endothermic reaction occurs, the heat required is absorbed from the thermal free energy of the solution, which decreases its temperature (Figure 1). The temperature change, along with the specific heat and mass of the solution, can then be used to summate the amount of estrus involved in either example.
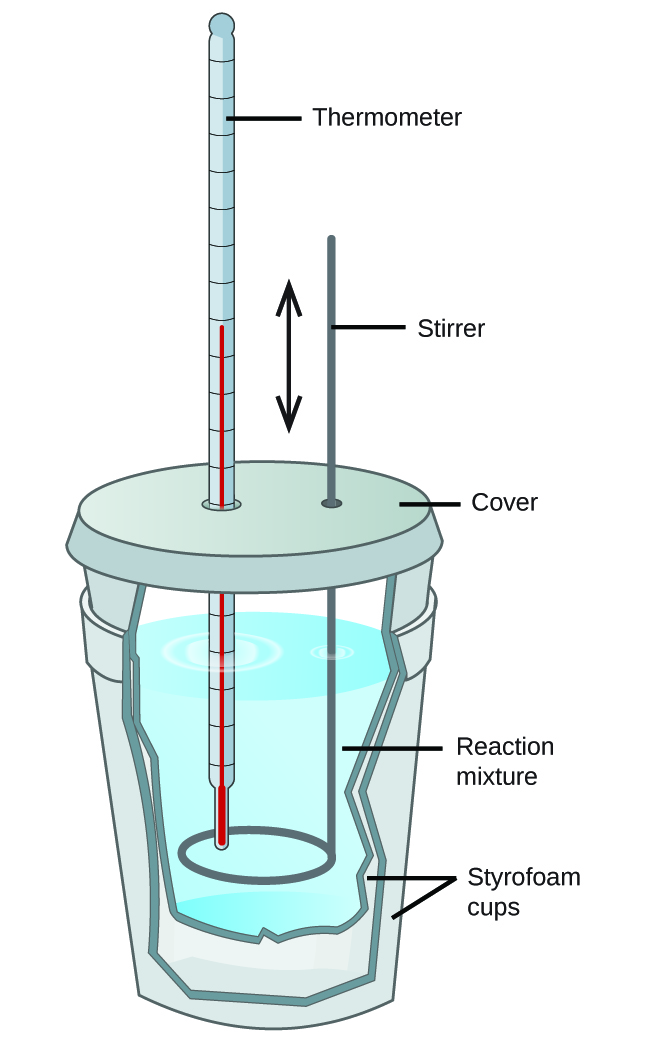
Scientists use well-insulated calorimeters that all but prevent the transfer of heat betwixt the calorimeter and its environment. This enables the authentic determination of the heat involved in chemical processes, the energy content of foods, and and so on. General chemical science students often employ simple calorimeters constructed from polystyrene cups (Effigy ii). These easy-to-utilise "coffee cup" calorimeters allow more heat commutation with their environs, and therefore produce less accurate free energy values.
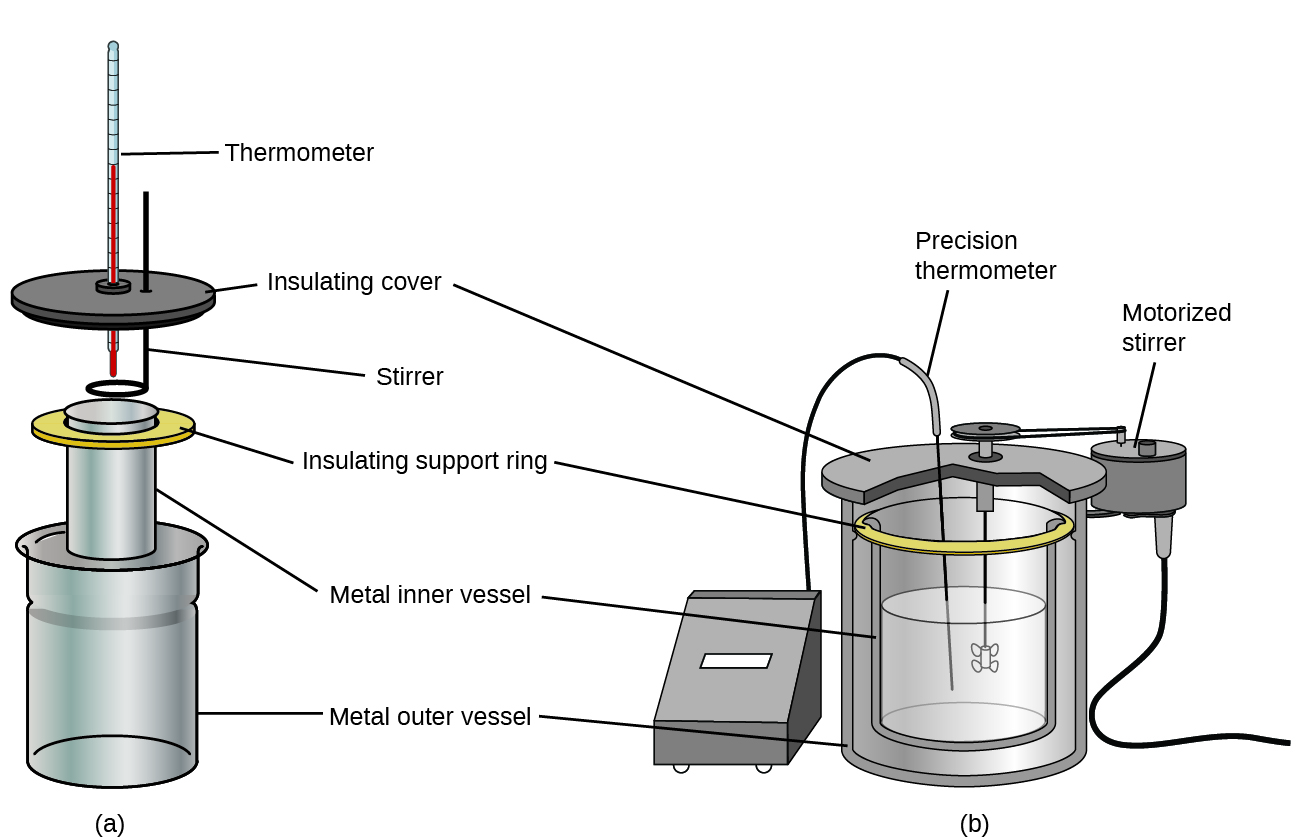
Commercial solution calorimeters are too available. Relatively cheap calorimeters often consist of two thin-walled cups that are nested in a way that minimizes thermal contact during use, along with an insulated cover, handheld stirrer, and simple thermometer. More than expensive calorimeters used for industry and inquiry typically take a well-insulated, fully enclosed reaction vessel, motorized stirring machinery, and a more authentic temperature sensor (Figure 3).
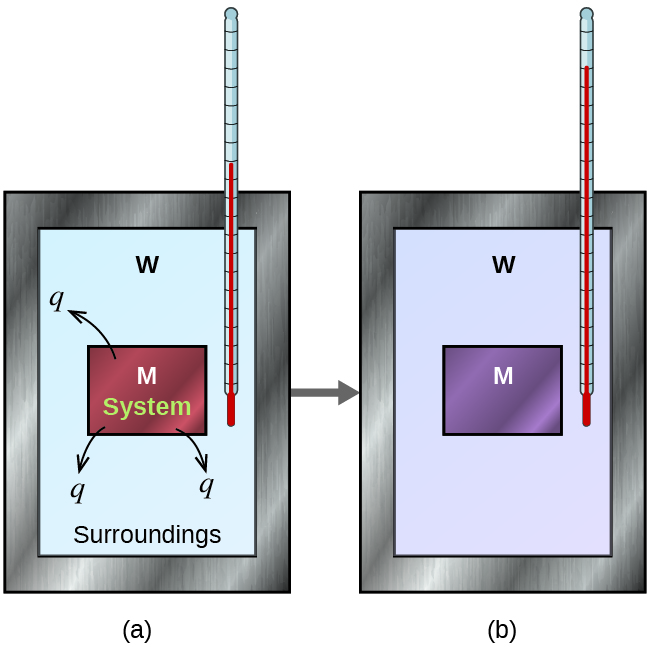
Earlier we practice calorimetry problems involving chemical reactions, consider a simpler example that illustrates the core thought behind calorimetry. Suppose nosotros initially accept a loftier-temperature substance, such as a hot piece of metal (M), and a depression-temperature substance, such equally cool water (Westward). If nosotros place the metal in the water, heat will flow from M to W. The temperature of M will decrease, and the temperature of Due west volition increment, until the two substances have the same temperature—that is, when they reach thermal equilibrium (Figure 4). If this occurs in a calorimeter, ideally all of this heat transfer occurs between the two substances, with no heat gained or lost past either the calorimeter or the calorimeter's environment. Under these ideal circumstances, the net oestrus change is null:
[latex]q_{\text{substance Yard}} + q_\text{substance W} = 0[/latex]
This relationship tin be rearranged to show that the rut gained by substance M is equal to the heat lost by substance Westward:
[latex]q_{\text{substance K}} = - q_{\text{substance W}}[/latex]
The magnitude of the heat (modify) is therefore the same for both substances, and the negative sign merely shows that q substance M and q substance W are opposite in management of heat flow (proceeds or loss) but does non indicate the arithmetic sign of either q value (that is determined by whether the matter in question gains or loses rut, per definition). In the specific situation described, q substance M is a negative value and q substance West is positive, since oestrus is transferred from M to W.
Example one
Heat Transfer between Substances at Different Temperatures
A 360-g piece of rebar (a steel rod used for reinforcing concrete) is dropped into 425 mL of h2o at 24.0 °C. The terminal temperature of the water was measured as 42.seven °C. Summate the initial temperature of the piece of rebar. Assume the specific heat of steel is approximately the same as that for iron (Table i in Chapter 5.1 Energy Nuts), and that all estrus transfer occurs betwixt the rebar and the water (in that location is no rut exchange with the surroundings).
Solution
The temperature of the h2o increases from 24.0 °C to 42.7 °C, so the water absorbs heat. That rut came from the piece of rebar, which initially was at a higher temperature. Assuming that all heat transfer was between the rebar and the h2o, with no estrus "lost" to the surroundings, then heat given off by rebar = −heat taken in by water, or:
[latex]q_{\text{rebar}} = -q_{\text{h2o}}[/latex]
Since we know how rut is related to other measurable quantities, nosotros have:
[latex](c \times m \times \Delta T)_{\text{rebar}} = -(c \times m \times \Delta T)_{\text{water}}[/latex]
Letting f = final and i = initial, in expanded form, this becomes:
[latex]c_{\text{rebar}} \times m_{\text{rebar}} \times (T_{\text{f, rebar}} - T_{\text{i, rebar}}) = -c_{\text{h2o}} \times m_{\text{water}} \times (T_{\text{f, water}} - T_{\text{i, water}})[/latex]
The density of h2o is 1.0 g/mL, so 425 mL of water = 425 g. Noting that the terminal temperature of both the rebar and water is 42.vii °C, substituting known values yields:
[latex](0.449 \;\text{J/one thousand} \;^\circ\text{C})(360 \;\text{g})(42.7 \;^\circ\text{C} - T_{\text{i, rebar}}) = (iv.184 \;\text{J/g} \;^\circ\text{C})(425 \;\text{g})(42.7 \;^\circ\text{C} - 24.0 \;^\circ\text{C})[/latex]
[latex]T_{\text{i, rebar}} = \frac{(4.184 \;\text{J/1000} \;^\circ\text{C})(425 \;\text{grand})(42.7 \;^\circ\text{C} - 24.0 \;^\circ\text{C})}{(0.449 \;\text{J/g} \;^\circ\text{C})(360 \;\text{g})} + 42.vii \;^\circ\text{C}[/latex]
Solving this gives T i,rebar= 248 °C, so the initial temperature of the rebar was 248 °C.
Check Your Learning
A 248-g piece of copper is dropped into 390 mL of water at 22.half dozen °C. The final temperature of the water was measured equally 39.nine °C. Calculate the initial temperature of the piece of copper. Assume that all estrus transfer occurs between the copper and the water.
Respond:
The initial temperature of the copper was 335.6 °C.
Check Your Learning
A 248-chiliad slice of copper initially at 314 °C is dropped into 390 mL of water initially at 22.6 °C. Assuming that all heat transfer occurs between the copper and the h2o, calculate the final temperature.
Answer:
The last temperature (reached by both copper and water) is 38.viii °C.
This method can too be used to determine other quantities, such as the specific oestrus of an unknown metal.
Instance 2
Identifying a Metallic by Measuring Specific Oestrus
A 59.7 thou slice of metallic that had been submerged in boiling water was chop-chop transferred into 60.0 mL of h2o initially at 22.0 °C. The concluding temperature is 28.5 °C. Use these data to determine the specific estrus of the metal. Employ this issue to identify the metallic.
Solution
Assuming perfect oestrus transfer, heat given off past metallic = −heat taken in past h2o, or:
[latex]q_{\text{metallic}} = -q_{\text{water}}[/latex]
In expanded class, this is:
[latex]c_{\text{metal}} \times m_{\text{metal}} \times (T_{\text{f, metal}} - T_{\text{i, metal}}) = -c_{\text{water}} \times m_{\text{water}} \times (T_{\text{f, h2o}} - T_{\text{i, h2o}})[/latex]
Noting that since the metal was submerged in humid water, its initial temperature was 100.0 °C; and that for water, 60.0 mL = threescore.0 g; nosotros take:
[latex](c_{\text{metal}}) (59.7 \;\text{thou}) (28.5 \;^\circ\text{C} - 100.0 \;^\circ\text{C}) = -(four.184 \;\text{J/1000} \;^\circ\text{C})(60.0 \;\text{g})(28.five \;^\circ\text{C} - 22.0 \;^\circ\text{C})[/latex]
Solving this:
[latex]c_{\text{metal}} = \frac{-(4.184 \;\text{J/1000} \;^\circ\text{C})(60.0 \;\text{yard})(6.5 \;^\circ\text{C})}{(59.7 \;\text{g})(-71.five \;^\circ\text{C})} = 0.38 \;\text{J/g} \;^\circ \text{C}[/latex]
Comparing this with values in Table 1 in Chapter 5.1 Energy Basics, our experimental specific heat is closest to the value for copper (0.39 J/thousand °C), so we place the metallic as copper.
Check Your Learning
A 92.9-k piece of a silver/gray metal is heated to 178.0 °C, and and so quickly transferred into 75.0 mL of water initially at 24.0 °C. Afterward 5 minutes, both the metal and the water have reached the aforementioned temperature: 29.7 °C. Determine the specific heat and the identity of the metallic. (Note: You should detect that the specific rut is shut to that of 2 different metals. Explain how yous can confidently determine the identity of the metallic).
Answer:
c metallic= 0.13 J/g °C
This specific heat is close to that of either gold or lead. It would exist difficult to determine which metal this was based solely on the numerical values. However, the observation that the metal is silver/greyness in addition to the value for the specific heat indicates that the metal is lead.
When we utilise calorimetry to determine the estrus involved in a chemical reaction, the same principles we have been discussing apply. The corporeality of heat captivated by the calorimeter is often small enough that we can neglect it (though non for highly accurate measurements, as discussed later), and the calorimeter minimizes energy exchange with the surround. Considering free energy is neither created nor destroyed during a chemical reaction, in that location is no overall energy change during the reaction. The heat produced or consumed in the reaction (the "arrangement"), q reaction, plus the heat captivated or lost by the solution (the "surround"), q solution, must add up to zero:
[latex]q_{\text{reaction}} + q_{\text{solution}} = 0[/latex]
This means that the amount of heat produced or consumed in the reaction equals the corporeality of heat absorbed or lost by the solution:
[latex]q_{\text{reaction}} = -q_{\text{solution}}[/latex]
This concept lies at the center of all calorimetry problems and calculations.
Example 3
Heat Produced by an Exothermic Reaction
When 50.0 mL of 0.ten M HCl(aq) and 50.0 mL of 0.10 M NaOH(aq), both at 22.0 °C, are added to a java cup calorimeter, the temperature of the mixture reaches a maximum of 28.9 °C. What is the estimate amount of estrus produced past this reaction?
[latex]\text{HCl}(aq) + \text{NaOH}(aq) \longrightarrow \text{NaCl}(aq) + \text{H}_2 \text{O}(l)[/latex]
Solution
To visualize what is going on, imagine that you could combine the 2 solutions then quickly that no reaction took place while they mixed; then later mixing, the reaction took place. At the instant of mixing, yous have 100.0 mL of a mixture of HCl and NaOH at 22.0 °C. The HCl and NaOH then react until the solution temperature reaches 28.nine °C.
The oestrus given off past the reaction is equal to that taken in by the solution. Therefore:
[latex]q_{\text{reaction}} = -q_{\text{solution}}[/latex]
(It is of import to remember that this relationship only holds if the calorimeter does not absorb any heat from the reaction, and there is no heat exchange between the calorimeter and its surroundings.)
Next, we know that the rut absorbed past the solution depends on its specific heat, mass, and temperature change:
[latex]q_{\text{solution}} = (c \times m \times \Delta T)_{\text{solution}}[/latex]
To proceed with this calculation, we need to make a few more reasonable assumptions or approximations. Since the solution is aqueous, nosotros can proceed equally if it were h2o in terms of its specific estrus and mass values. The density of water is approximately one.0 one thousand/mL, and so 100.0 mL has a mass of nearly i.0 × x2 g (two significant figures). The specific heat of water is approximately 4.18 J/g °C, so we utilise that for the specific heat of the solution. Substituting these values gives:
[latex]q_{\text{solution}} = (4.184 \;\text{J/g} \;^\circ\text{C})(1.0 \times 10^2 \;\text{g})(28.nine \;^\circ\text{C} - 22.0 \;^\circ\text{C}) = two.89 \times 10^three \;\text{J}[/latex]
Finally, since nosotros are trying to find the heat of the reaction, we have:
[latex]q_{\text{reaction}} = -q_{\text{solution}} = -2.89 \times 10^3 \;\text{J}[/latex]
The negative sign indicates that the reaction is exothermic. It produces ii.89 kJ of heat.
Check Your Learning
When 100 mL of 0.200 1000 NaCl(aq) and 100 mL of 0.200 M AgNOthree(aq), both at 21.9 °C, are mixed in a java cup calorimeter, the temperature increases to 23.5 °C as solid AgCl forms. How much heat is produced by this precipitation reaction? What assumptions did you brand to decide your value?
Answer:
1.34 × 103 J; assume no heat is absorbed past the calorimeter, no rut is exchanged betwixt the calorimeter and its environs, and that the specific heat and mass of the solution are the aforementioned as those for water
Thermochemistry of Mitt Warmers
When working or playing outdoors on a cold day, you might utilize a hand warmer to warm your hands (Figure five). A common reusable manus warmer contains a supersaturated solution of NaC2HiiiO2 (sodium acetate) and a metallic disc. Bending the disk creates nucleation sites around which the metastable NaCtwoHiiiO2 quickly crystallizes (a later chapter on solutions will investigate saturation and supersaturation in more detail).
The process [latex]\text{NaC}_2 \text{H}_3 \text{O}_2 (aq) \longrightarrow \text{NaC}_2 \text{H}_3 \text{O}_2 (s)[/latex] is exothermic, and the rut produced past this process is absorbed by your hands, thereby warming them (at least for a while). If the hand warmer is reheated, the NaCtwoH3Oii redissolves and can exist reused.

Another common paw warmer produces heat when it is ripped open up, exposing iron and water in the hand warmer to oxygen in the air. One simplified version of this exothermic reaction is [latex]2\text{Iron}(s) + \frac{three}{2}\text{O}_2(1000) \longrightarrow \text{Atomic number 26}_2\text{O}_3(southward)[/latex]. Table salt in the hand warmer catalyzes the reaction, so it produces heat more chop-chop; cellulose, vermiculite, and activated carbon aid distribute the estrus evenly. Other types of mitt warmers employ lighter fluid (a platinum catalyst helps lighter fluid oxidize exothermically), charcoal (charcoal oxidizes in a special case), or electrical units that produce estrus by passing an electrical current from a bombardment through resistive wires.

This link shows the atmospheric precipitation reaction that occurs when the deejay in a chemic hand warmer is flexed.
Example 4
Estrus Flow in an Instant Ice Pack
When solid ammonium nitrate dissolves in water, the solution becomes cold. This is the basis for an "instant ice pack" (Effigy vi). When 3.21 g of solid NH4NOthree dissolves in 50.0 g of water at 24.9 °C in a calorimeter, the temperature decreases to twenty.3 °C.
Calculate the value of q for this reaction and explain the meaning of its arithmetic sign. State any assumptions that you lot made.

Solution
Nosotros assume that the calorimeter prevents heat transfer between the solution and its external environment (including the calorimeter itself), in which case:
[latex]q_{\text{rxn}} = -q_{\text{soln}}[/latex]
with "rxn" and "soln" used as shorthand for "reaction" and "solution," respectively.
Assuming likewise that the specific heat of the solution is the aforementioned as that for water, we have:
[latex]\begin{array}{r @{{}={}} l} q_{\text{rxn}} & -{q_{\text{soln}}} = -(c \times g \times \Delta T)_{\text{soln}} \\[1em] & -[(4.184 \;\text{J/g} \;^\circ\text{C}) \times (53.two \;\text{g}) \times (20.3 \;^\circ\text{C} - 24.9 \;^\circ\text{C})] \\[1em] & -[(4.184 \;\text{J/g} \;^\circ\text{C}) \times (53.two \;\text{g}) \times (-iv.6 \;^\circ\text{C})] \\[1em] & + 1.0 \times 10^3 \;\text{J} = +1.0 \;\text{kJ} \terminate{array}[/latex]
The positive sign for q indicates that the dissolution is an endothermic procedure.
Check Your Learning
When a 3.00-thousand sample of KCl was added to 3.00 × 102 thousand of water in a coffee cup calorimeter, the temperature decreased by 1.05 °C. How much estrus is involved in the dissolution of the KCl? What assumptions did you make?
Answer:
1.33 kJ; assume that the calorimeter prevents heat transfer between the solution and its external environs (including the calorimeter itself) and that the specific oestrus of the solution is the aforementioned as that for h2o
If the amount of estrus captivated past a calorimeter is besides large to fail or if we require more accurate results, then we must take into account the oestrus captivated both by the solution and by the calorimeter.
The calorimeters described are designed to operate at abiding (atmospheric) force per unit area and are user-friendly to measure heat flow accompanying processes that occur in solution. A unlike blazon of calorimeter that operates at constant book, colloquially known as a bomb calorimeter, is used to measure the energy produced past reactions that yield big amounts of heat and gaseous products, such as combustion reactions. (The term "bomb" comes from the ascertainment that these reactions can be vigorous enough to resemble explosions that would damage other calorimeters.) This type of calorimeter consists of a robust steel container (the "bomb") that contains the reactants and is itself submerged in water (Figure vii). The sample is placed in the bomb, which is and so filled with oxygen at high pressure. A small electrical spark is used to ignite the sample. The free energy produced past the reaction is trapped in the steel flop and the surrounding water. The temperature increment is measured and, forth with the known heat capacity of the calorimeter, is used to calculate the energy produced past the reaction. Bomb calorimeters crave calibration to determine the heat chapters of the calorimeter and ensure accurate results. The calibration is achieved using a reaction with a known q, such as a measured quantity of benzoic acrid ignited by a spark from a nickel fuse wire that is weighed before and after the reaction. The temperature change produced by the known reaction is used to determine the estrus capacity of the calorimeter. The scale is by and large performed each time before the calorimeter is used to assemble research data.


Click on this link to view how a bomb calorimeter is prepared for action.
This site shows calorimetric calculations using sample information.
Example 5
Bomb Calorimetry
When 3.12 g of glucose, Chalf dozenH12O6, is burned in a flop calorimeter, the temperature of the calorimeter increases from 23.8 °C to 35.six °C. The calorimeter contains 775 g of water, and the bomb itself has a heat capacity of 893 J/°C. How much rut was produced by the combustion of the glucose sample?
Solution
The combustion produces oestrus that is primarily absorbed by the h2o and the bomb. (The amounts of heat absorbed past the reaction products and the unreacted backlog oxygen are relatively modest and dealing with them is beyond the scope of this text. Nosotros will neglect them in our calculations.)
The heat produced by the reaction is absorbed by the water and the flop:
[latex]\brainstorm{array}{r @{{}={}} l} q_{\text{rxn}} & -({q_{\text{water}} + q_{\text{bomb}}}) \\[1em] & -[(four.184 \;\text{J/g} \;^\circ\text{C}) \times (775 \;\text{yard}) \times (35.half dozen \;^\circ\text{C} - 23.8 \;^\circ\text{C}) + 893\;\text{J/}^\circ\text{C} \times (35.6 \;^\circ\text{C} - 23.8 \;^\circ\text{C})] \\[1em] & -(38,300 \;\text{J} + ten,500 \;\text{J}) \\[1em] & -48,800 \;\text{J} = -48.8 \;\text{kJ} \finish{array}[/latex]
This reaction released 48.7 kJ of oestrus when 3.12 1000 of glucose was burned.
Check Your Learning
When 0.963 chiliad of benzene, C6H6, is burned in a bomb calorimeter, the temperature of the calorimeter increases by 8.39 °C. The bomb has a heat capacity of 784 J/°C and is submerged in 925 mL of h2o. How much heat was produced by the combustion of the glucose sample?
Since the first 1 was constructed in 1899, 35 calorimeters have been built to measure the oestrus produced by a living person.[1] These whole-body calorimeters of various designs are large enough to concur an individual human being. More recently, whole-room calorimeters allow for relatively normal activities to be performed, and these calorimeters generate data that more than closely reflect the real world. These calorimeters are used to measure the metabolism of individuals under unlike ecology conditions, different dietary regimes, and with different health conditions, such as diabetes. In humans, metabolism is typically measured in Calories per day. A nutritional calorie (Calorie) is the energy unit of measurement used to quantify the amount of energy derived from the metabolism of foods; one Calorie is equal to one thousand calories (1 kcal), the amount of energy needed to heat i kg of water by 1 °C.
Measuring Nutritional Calories
In your day-to-mean solar day life, you may be more familiar with energy being given in Calories, or nutritional calories, which are used to quantify the corporeality of free energy in foods. One calorie (cal) = exactly 4.184 joules, and i Calorie (note the capitalization) = 1000 cal, or i kcal. (This is approximately the amount of energy needed to heat one kg of h2o past one °C.)
The macronutrients in nutrient are proteins, carbohydrates, and fats or oils. Proteins provide about 4 Calories per gram, carbohydrates as well provide about iv Calories per gram, and fats and oils provide about ix Calories/m. Nutritional labels on food packages show the caloric content of one serving of the food, as well every bit the breakup into Calories from each of the three macronutrients (Figure viii).
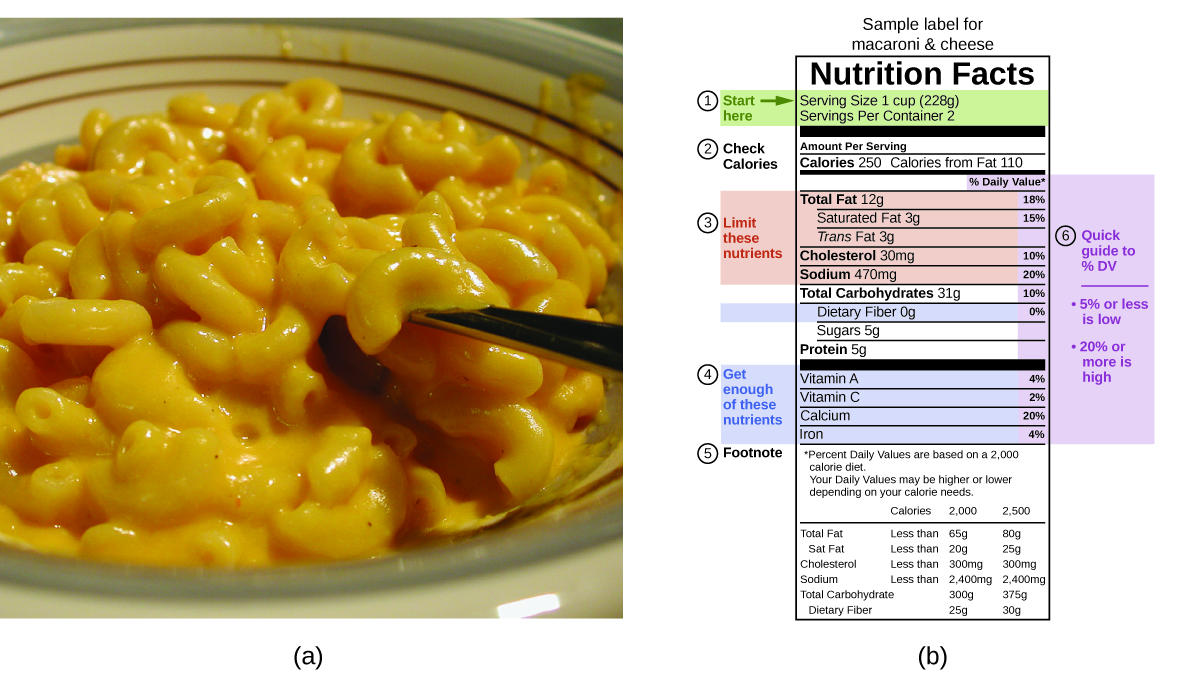
For the instance shown in (b), the total energy per 228-g portion is calculated past:
[latex](v \;\text{g protein} \times four \;\text{Calories/g}) + (31 \;\text{g carb} \times 4 \;\text{Calories/g}) + (12 \;\text{k fatty} \times 9 \;\text{Calories/g}) = 252 \;\text{Calories}[/latex]
And so, yous can use food labels to count your Calories. Merely where do the values come from? And how authentic are they? The caloric content of foods tin can be adamant by using bomb calorimetry; that is, by burning the food and measuring the free energy it contains. A sample of nutrient is weighed, mixed in a blender, freeze-dried, ground into pulverisation, and formed into a pellet. The pellet is burned inside a bomb calorimeter, and the measured temperature change is converted into free energy per gram of food.
Today, the caloric content on food labels is derived using a method called the Atwater system that uses the average caloric content of the different chemical constituents of food, poly peptide, saccharide, and fats. The average amounts are those given in the equation and are derived from the diverse results given by bomb calorimetry of whole foods. The sugar corporeality is discounted a sure amount for the fiber content, which is indigestible saccharide. To make up one's mind the energy content of a food, the quantities of carbohydrate, poly peptide, and fat are each multiplied by the average Calories per gram for each and the products summed to obtain the total free energy.

Click on this link to access the US Department of Agronomics (USDA) National Nutrient Database, containing nutritional information on over 8000 foods.
Central Concepts and Summary
Calorimetry is used to measure the amount of thermal energy transferred in a chemical or physical process. This requires careful measurement of the temperature modify that occurs during the process and the masses of the system and surroundings. These measured quantities are and then used to compute the amount of heat produced or consumed in the process using known mathematical relations.
Calorimeters are designed to minimize energy exchange between the system beingness studied and its environs. They range from simple coffee cup calorimeters used by introductory chemistry students to sophisticated flop calorimeters used to decide the free energy content of food.
Chemistry End of Chapter Exercises
- A 500-mL bottle of water at room temperature and a two-L bottle of water at the same temperature were placed in a fridge. Later 30 minutes, the 500-mL bottle of water had cooled to the temperature of the refrigerator. An hr later, the 2-50 of water had cooled to the same temperature. When asked which sample of water lost the most heat, one educatee replied that both bottles lost the aforementioned corporeality of rut because they started at the aforementioned temperature and finished at the same temperature. A second student thought that the 2-L bottle of water lost more heat because there was more water. A tertiary educatee believed that the 500-mL bottle of water lost more than heat because it cooled more than speedily. A 4th educatee thought that it was not possible to tell because we do not know the initial temperature and the terminal temperature of the water. Bespeak which of these answers is correct and describe the mistake in each of the other answers.
- Would the amount of heat measured for the reaction in Example 3 be greater, lesser, or remain the aforementioned if we used a calorimeter that was a poorer insulator than a java cup calorimeter? Explain your answer.
- Would the corporeality of heat absorbed past the dissolution in Example four announced greater, lesser, or remain the same if the experimenter used a calorimeter that was a poorer insulator than a coffee cup calorimeter? Explain your answer.
- Would the amount of heat absorbed past the dissolution in Example 4 appear greater, lesser, or remain the same if the oestrus capacity of the calorimeter were taken into account? Explicate your reply.
- How many milliliters of h2o at 23 °C with a density of i.00 k/mL must exist mixed with 180 mL (nearly 6 oz) of coffee at 95 °C so that the resulting combination volition have a temperature of 60 °C? Presume that java and water have the same density and the aforementioned specific heat.
- How much volition the temperature of a cup (180 g) of coffee at 95 °C be reduced when a 45 g silvery spoon (specific heat 0.24 J/g °C) at 25 °C is placed in the coffee and the two are allowed to reach the aforementioned temperature? Assume that the coffee has the same density and specific heat equally water.
- A 45-g aluminum spoon (specific heat 0.88 J/g °C) at 24 °C is placed in 180 mL (180 k) of coffee at 85 °C and the temperature of the two go equal.
(a) What is the final temperature when the two become equal? Assume that java has the same specific heat every bit h2o.
(b) The first time a educatee solved this problem she got an answer of 88 °C. Explain why this is clearly an wrong answer.
- The temperature of the cooling h2o every bit it leaves the hot engine of an automobile is 240 °F. After it passes through the radiator information technology has a temperature of 175 °F. Calculate the corporeality of heat transferred from the engine to the surroundings by one gallon of water with a specific heat of 4.184 J/g °C.
- A 70.0-chiliad piece of metal at fourscore.0 °C is placed in 100 1000 of water at 22.0 °C contained in a calorimeter like that shown in Effigy 2. The metal and water come up to the same temperature at 24.half dozen °C. How much heat did the metal give up to the water? What is the specific heat of the metal?
- If a reaction produces 1.506 kJ of oestrus, which is trapped in thirty.0 chiliad of water initially at 26.5 °C in a calorimeter like that in Figure 2, what is the resulting temperature of the water?
- A 0.500-g sample of KCl is added to l.0 g of water in a calorimeter (Figure 2). If the temperature decreases by 1.05 °C, what is the approximate amount of heat involved in the dissolution of the KCl, bold the heat capacity of the resulting solution is four.18 J/chiliad °C? Is the reaction exothermic or endothermic?
- Dissolving iii.0 g of CaCl2(due south) in 150.0 g of water in a calorimeter (Figure 2) at 22.iv °C causes the temperature to ascent to 25.8 °C. What is the approximate corporeality of heat involved in the dissolution, assuming the heat capacity of the resulting solution is 4.18 J/g °C? Is the reaction exothermic or endothermic?
- When 50.0 one thousand of 0.200 M NaCl(aq) at 24.1 °C is added to 100.0 g of 0.100 Chiliad AgNO3(aq) at 24.i °C in a calorimeter, the temperature increases to 25.2 °C equally AgCl(s) forms. Assuming the specific rut of the solution and products is iv.xx J/1000 °C, calculate the judge amount of rut in joules produced.
- The improver of 3.xv g of Ba(OH)2·8H2O to a solution of 1.52 grand of NH4SCN in 100 one thousand of water in a calorimeter caused the temperature to fall by 3.1 °C. Bold the specific estrus of the solution and products is 4.twenty J/chiliad °C, calculate the approximate amount of heat absorbed by the reaction, which can be represented past the following equation:
Ba(OH)2·8HtwoO(due south) + 2NH4SCN(aq) ⟶ Ba(SCN)2(aq) + 2NH3(aq) + 10H2O(l)
- The reaction of fifty mL of acid and fifty mL of base described in Instance 3 increased the temperature of the solution by 6.9 degrees. How much would the temperature accept increased if 100 mL of acid and 100 mL of base of operations had been used in the same calorimeter starting at the same temperature of 22.0 °C? Explain your answer.
- If the 3.21 g of NH4NO3 in Case four were dissolved in 100.0 g of water under the same conditions, how much would the temperature alter? Explain your respond.
- When 1.0 1000 of fructose, C6H12O6(southward), a sugar commonly found in fruits, is burned in oxygen in a bomb calorimeter, the temperature of the calorimeter increases past 1.58 °C. If the heat capacity of the calorimeter and its contents is 9.90 kJ/°C, what is q for this combustion?
- When a 0.740-g sample of trinitrotoluene (TNT), C7HvDue north2O6, is burned in a bomb calorimeter, the temperature increases from 23.4 °C to 26.9 °C. The heat capacity of the calorimeter is 534 J/°C, and information technology contains 675 mL of water. How much heat was produced by the combustion of the TNT sample?
- One method of generating electricity is past burning coal to heat h2o, which produces steam that drives an electric generator. To make up one's mind the rate at which coal is to be fed into the burner in this type of plant, the heat of combustion per ton of coal must be determined using a flop calorimeter. When 1.00 k of coal is burned in a bomb calorimeter (Figure 7), the temperature increases by 1.48 °C. If the heat capacity of the calorimeter is 21.vi kJ/°C, determine the oestrus produced by combustion of a ton of coal (2.000 × 103 pounds).
- The corporeality of fatty recommended for someone with a daily nutrition of 2000 Calories is 65 m. What per centum of the calories in this diet would be supplied by this amount of fatty if the average number of Calories for fat is ix.1 Calories/g?
- A teaspoon of the carbohydrate sucrose (common sugar) contains 16 Calories (16 kcal). What is the mass of ane teaspoon of sucrose if the average number of Calories for carbohydrates is 4.1 Calories/m?
- What is the maximum mass of carbohydrate in a 6-oz serving of diet soda that contains less than i Calorie per can if the boilerplate number of Calories for carbohydrates is four.1 Calories/thou?
- A pint of premium ice cream can incorporate 1100 Calories. What mass of fatty, in grams and pounds, must be produced in the trunk to store an extra 1.ane × 103 Calories if the average number of Calories for fatty is 9.1 Calories/thousand?
- A serving of a breakfast cereal contains 3 m of protein, 18 grand of carbohydrates, and half dozen g of fat. What is the Calorie content of a serving of this cereal if the average number of Calories for fat is 9.1 Calories/g, for carbohydrates is 4.1 Calories/g, and for protein is 4.one Calories/g?
- Which is the to the lowest degree expensive source of energy in kilojoules per dollar: a box of breakfast cereal that weighs 32 ounces and costs $4.23, or a liter of isooctane (density, 0.6919 g/mL) that costs $0.45? Compare the nutritional value of the cereal with the estrus produced by combustion of the isooctane under standard conditions. A ane.0-ounce serving of the cereal provides 130 Calories.
Glossary
- bomb calorimeter
- device designed to measure the energy change for processes occurring under weather of abiding volume; commonly used for reactions involving solid and gaseous reactants or products
- calorimeter
- device used to measure the amount of heat captivated or released in a chemical or concrete process
- calorimetry
- procedure of measuring the amount of rut involved in a chemic or concrete procedure
- nutritional calorie (Calorie)
- unit of measurement used for quantifying free energy provided by digestion of foods, defined as m cal or ane kcal
- surroundings
- all matter other than the system existence studied
- organization
- portion of matter undergoing a chemic or physical modify existence studied
Solutions
Answers to Chemistry End of Chapter Exercises
2. lesser; more than heat would be lost to the coffee cup and the environment and so ΔT for the water would be lesser and the calculated q would be lesser
4. greater, since taking the calorimeter's heat capacity into account volition compensate for the thermal energy transferred to the solution from the calorimeter; this approach includes the calorimeter itself, along with the solution, as "surroundings": q rxn = −(q solution + q calorimeter); since both q solution and q calorimeter are negative, including the latter term (q rxn) will yield a greater value for the heat of the dissolution
six. The temperature of the java will drop 1 degree.
viii. five.7 × 102 kJ
10. 38.5 °C
12. 2.ii kJ; The oestrus produced shows that the reaction is exothermic.
14. 1.4 kJ
16. 22.six. Since the mass and the heat capacity of the solution is approximately equal to that of the water, the 2-fold increment in the corporeality of water leads to a two-fold decrease of the temperature modify.
xviii. 11.vii kJ
20. 30%
22. 0.24 g
24. 1.four × x2 Calories
Source: https://opentextbc.ca/chemistry/chapter/5-2-calorimetry/
Post a Comment for "What Is the Amount of Heat Required to Raise the Temperature of 200.0 G of Aluminum by 10ã‚â°c"